What the FDA Still Doesn't Know About Estrogen and Males
A citizen petition backed by research, detransitioners, and a dozen organizations is asking the FDA to do its job. Add your comment to strengthen the case and tip the scales.
In December 2025, a coalition of parents, researchers, clinicians, and advocacy organizations -- including the LGB Courage Coalition -- filed a citizen petition with the Food and Drug Administration. The request was straightforward: look at the evidence on what long-term, high-dose estrogen does to male bodies, and act accordingly. (The petition is available for download here, under the Attachments section.)
The FDA has 180 days to respond. That clock runs out in June 2026. The public comment portal is open right now. Your comments could help shape whether the FDA takes this petition seriously. In this article, we discuss what the petition demands, why the evidence backing it is hard to dismiss, and how you can comment effectively.
A practice built for one population, now applied to another
Estrogen has been prescribed to males as part of gender medicine since at least the 1990s. Back then, the patient group was small, mostly older adults seeking palliative relief from acute distress, and they often had competing health conditions that shifted the calculus for long-term risks.
The current cohort is no longer a small group of older adults. It now consists of many adolescents and young adults who begin estrogen therapy early and anticipate continued, lifelong use. The safety protocols have not kept pace. Clinicians continue to use short-term measurement tools -- clinical studies limited to 2-5-year observation windows -- to represent lifetime outcomes.
Estrogen does not reduce risk of suicide
One of the primary justifications of cross-sex hormone treatment, across all ages, is that it improves mental health and reduces suicidality. Proponents have argued for years that reducing the risk of suicide justifies any number of other medical risks. However, the data shows just the opposite — use of estrogen for gender affirming purposes may actually increase the risk of suicide.
One study cited in the petition, a re-analysis by Michael Biggs of results originally produced by Jack Turban, found that “males who took estrogen are more likely to plan suicide, to attempt suicide, and to require hospitalization for a suicide attempts.”
Another study cited in the petition found that the most common adverse event reported by patients after starting estrogen was suicidal ideation. Suicide prevention cannot be claimed as a proven beneficial outcome.
What cumulative estrogen exposure does to men
The petition draws on a growing body of peer-reviewed evidence showing what cumulative estrogen exposure can do to male physiology over time:
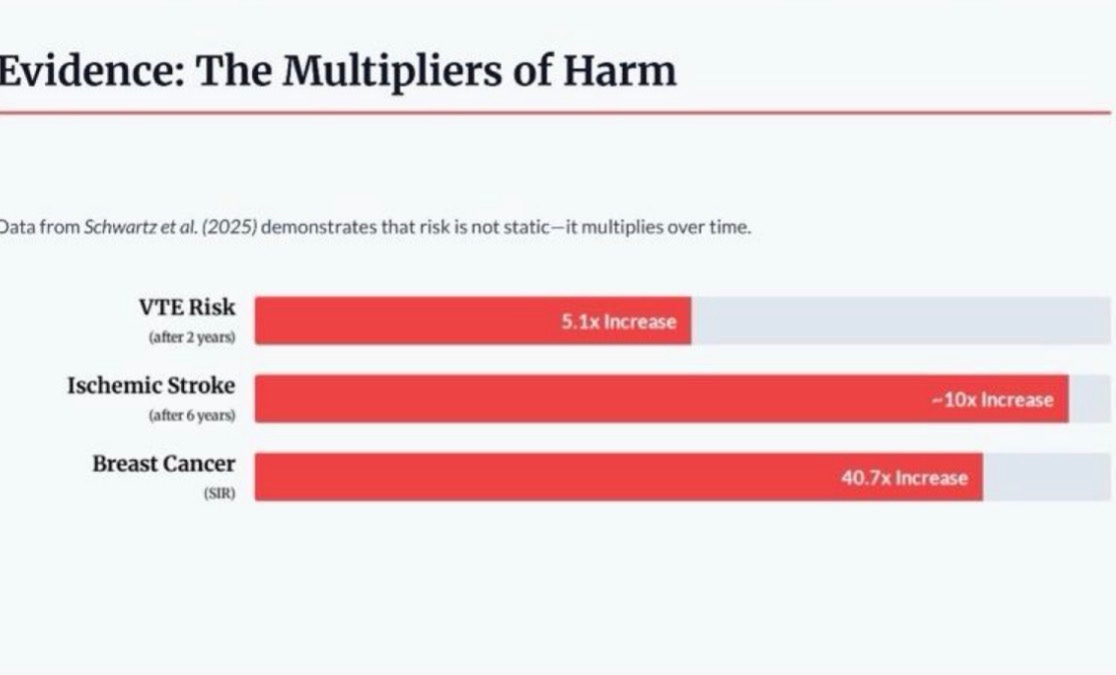
Venous thromboembolism (VTE) risk increases 5.1 times after just two years of exogenous estrogen use.
Ischemic stroke risk increases approximately 10 times after six years.
Breast cancer incidence, measured by standardized incidence ratio, increases 40.7 times.
Risk multipliers over time (Schwartz et al., 2025): VTE, ischemic stroke, and breast cancer incidence in trans-identified males on long-term estrogen. Risks are not static -- they accumulate and escalate with duration of exposure.
These are not weak signals. They are numbers that, in any other pharmacological context, would trigger immediate regulatory review.
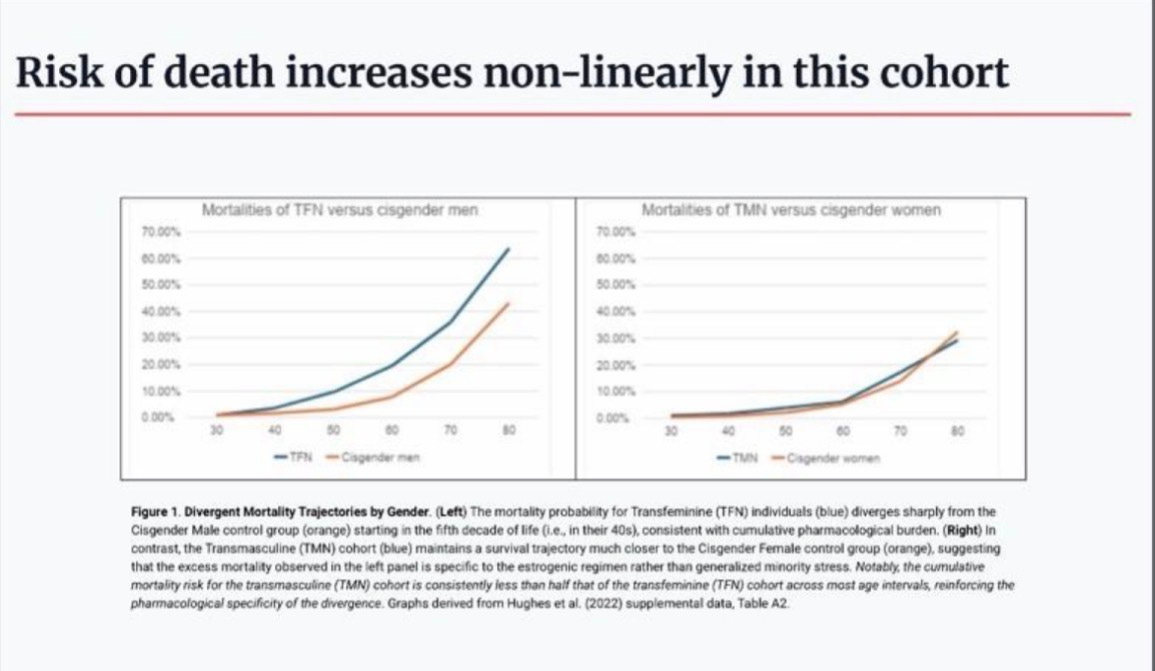
Cumulative effects are shown in the overall mortality data cited in the petition. Mortality rates for males on estrogen start to diverge from the general male population significantly after age 30, and climb more than three times higher by the 50s. Notably, trans-identified females do not show the same mortality divergence from other women of similar age, which suggests the excess mortality is more likely attributable to the estrogen itself, than to some general “minority stress” effect.
Divergent mortality trajectories (Hughes et al., 2022, Table A2): The mortality probability for trans-identified males (blue, left panel) diverges sharply from male controls (orange) starting in the fifth decade of life. Trans-identified females (right panel) maintain survival trajectories much closer to female controls, reinforcing that the divergence is pharmacologically specific.
In short, growing evidence suggests that estrogen, when used for gender affirming purposes in men, does not reduce suicidality and shows the potential for real physical harm. Patients are being told a treatment is safe and effective, yet the data not only fail to support those claims, they appear to actively contradict them. This situation falls into the FDA’s mandate to ensure that drugs prescribed by doctors are safe, effective, and properly labelled.
What the petition asks the FDA to do
The petition makes three core requests. First, a mandatory boxed warning on estrogen packaging -- the strongest label the FDA can require -- explicitly naming stroke, cancer, sterility, and cognitive decline as documented risks. Second, a public hearing under Part 15 of FDA regulations, giving detransitioners, clinical experts, and affected families the opportunity to testify on the record. Third, a Risk Evaluation and Mitigation Strategy (REMS) and mandatory patient registry, requiring post-market surveillance over a minimum of 20 years rather than relying on the short-term studies that currently shape clinical guidelines.
Three actions recommended in the petition: a mandatory boxed warning, a public “Part 15” hearing, and a REMS evaluation with long-term patient registry.
These are the agency’s standard tools when serious harm signals emerge around a widely prescribed drug. That this level of review and safeguarding was not implemented years ago defies understanding.
How to make your comment matter
While every comment counts, the FDA is not only counting votes but collecting data. Relevant observations -- a parent describing what their son was not told before starting treatment, a detransitioner documenting a specific diagnosis they received, a clinician noting what they observed in practice -- can really influence the FDA.
Commenting is easy. Just keep it simple. You can choose to remain anonymous, but do identify yourself as a parent, detransitioner, clinician, researcher, friend or family member of someone affected, or simply a concerned citizen. Then state clearly that you support the petition and specify what you are asking for:
A mandatory boxed warning under 21 C.F.R. § 201.80(e) naming stroke, cancer, sterility, and cognitive decline
A “Part 15” public hearing to get transparent scientific debate on the record
A comprehensive safety review and REMS evaluation
Mandatory enhanced adverse event reporting
Development of communications for medical providers regarding safeguards
Lastly, describe what you witnessed or experienced: specific diagnoses, what you were told versus what happened, whether you were told about the risks, details that the information you were given at the time missed, but your own experience did not.
(You can read other comments here.)
Where to submit your comment
The docket is active until the FDA acts, with a current horizon of June 2026. Submit your comment here -- be sure to use this specific link. Other pages on the site may not display the comment option correctly.
The LGB Courage Coalition co-signed this petition because we believe the FDA has both the authority and the obligation to act when a widely prescribed drug causes serious harm which the medical establishment has failed to either acknowledge or mitigate. The evidence is substantial. We must provide the public record of harm to urge the FDA to act.
You can help build that record. The comment window is open. Use it.